 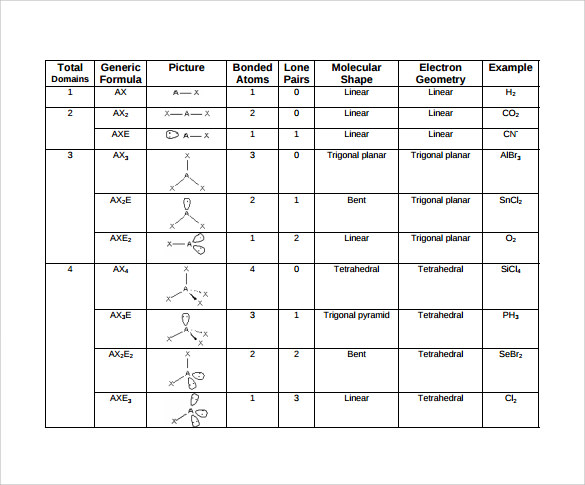
It is based on the idea that atoms in a molecule will share or transfer electrons in order to achieve a stable electron configuration, known as an octet. Lewis theory is a method of representing the valence electrons of an atom in a molecule. Lewis Theory and The Valence-Shell Electron-Pair Repulsion Theory In a nonpolar molecule, the atoms have similar electronegativities, creating an evenly distributed distribution of electrons. In a polar molecule, the difference in electronegativity of the atoms causes the electrons to be distributed unevenly, creating a partial positive charge on one end of the molecule and a partial negative charge on the other. Fluorine (F) has the highest electronegativity of all elements, while Francium (Fr) has the lowest. Atoms with a high electronegativity will tend to pull electrons closer to themselves, while atoms with a low electronegativity will tend to release electrons. Methane (CH4) is a common example of a nonpolar molecule.Įlectronegativity is a measure of an atom’s ability to attract electrons to itself. 
This can occur when the atoms in a molecule have similar electronegativities, creating an evenly distributed distribution of electrons. Water (H2O) is a common example of a polar molecule.Ī nonpolar molecule is a molecule that has a symmetrical distribution of electrons, so there is no net dipole moment. This can occur when the atoms in a molecule have different electronegativities, creating a partial positive charge on one end of the molecule and a partial negative charge on the other. In some cases, the electrons in a molecule may be arranged differently than the atoms.Ĭommon Molecular Structures Polar MoleculesĪ polar molecule is a molecule that has a net dipole moment, meaning that the positive and negative charges are not evenly distributed. It’s worth noting that molecular geometry is not always the same as electron geometry.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
